FDA Recall: Webcol™ Large Alcohol Prep Pads Contaminated With Paenibacillus phoenicis
Cardinal Health has issued a voluntary, nationwide recall of specific lots of Webcol™ Large Alcohol Prep Pads (70 % isopropyl alcohol). Routine quality testing detected contamination with Paenibacillus phoenicis, an environmental bacterium capable of causing serious skin or bloodstream infections—particularly in people with weakened immune systems. If you stock or use these pads, locate the lot number immediately, quarantine affected stock, and follow the return instructions below.
Key Recall Facts
- Recall initiation: March 19, 2024 (FDA posted March 20, 2024)
- Product: Webcol™ Large Alcohol Prep Pads (70 % isopropyl alcohol)
- Reason: Microbial contamination with P. phoenicis
- Distribution period: Sept 2023 – Feb 2024 (U.S., Puerto Rico, Japan)
- Manufacturer: Cardinal Health | Dublin, Ohio
Sources
Health Risks From Contaminated Webcol™ Pads
Alcohol prep pads are used to disinfect the skin before injections and minor procedures. A contaminated pad can transfer bacteria directly into the body. High-risk groups—such as chemotherapy patients, transplant recipients, or anyone with poorly controlled diabetes—could develop:
- Localized skin or soft-tissue infections
- Bacteremia (bloodstream infection)
- Central-nervous-system infections
- Sepsis or other life-threatening complications
How to Identify Recalled Webcol™ Alcohol Prep Pads
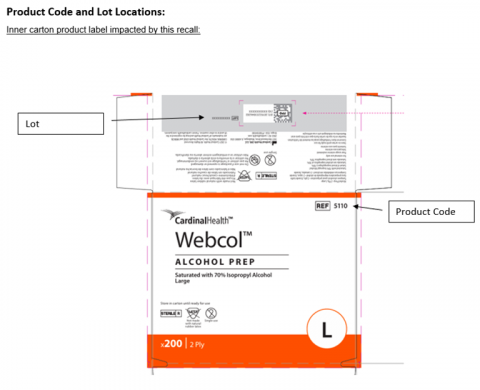
The affected lot numbers are listed in the PDF recall notice (see button above). Lot numbers and product code appear on both the inner carton and outer case labels, as illustrated in the FDA images below.
Immediate Steps If You Have Webcol™ Large Alcohol Prep Pads
- Stop using any Webcol™ Large Alcohol Prep Pads until you verify the lot is not recalled.
- Quarantine & label products from recalled lots to prevent accidental use.
- Contact Cardinal Health to acknowledge the recall and arrange returns:
- Email: GMB-FieldCorrectiveAction@cardinalhealth.com
- Phone: 1-800-292-9332 (Mon–Fri, 8 am – 5 pm ET)
- Fax the acknowledgement form to 614-652-9648
- Notify downstream customers if you redistributed the product.
- Report adverse events to the FDA MedWatch program:
- MedWatch Online Reporting
- Phone: 1-800-332-1088
Recall Images From FDA

Frequently Asked Questions
Why are Webcol™ Large Alcohol Prep Pads being recalled?
Routine quality testing discovered contamination with Paenibacillus phoenicis, which can cause infections if introduced into the skin or bloodstream.
Which lot numbers of Webcol™ Large Alcohol Prep Pads are affected?
All recalled lot numbers appear in the official PDF notice. Compare the lot on your carton or case to that list.
What should I do if I have Webcol™ Large Alcohol Prep Pads from a recalled lot?
Immediately stop using the pads, isolate the inventory, and contact Cardinal Health to coordinate returns or disposal.
Are Webcol™ Large Alcohol Prep Pads from non-recalled lots safe?
Yes. Cardinal Health and FDA state that only the specific lots listed in the recall are affected. Always verify the lot number before use.
How do I obtain a refund or replacement for recalled Webcol™ Large Alcohol Prep Pads?
Fill out the recall acknowledgement form included in Cardinal Health’s notice, then email or fax the form to the company. They will arrange credit or replacement product.
Who can I contact with questions about the Webcol™ Large Alcohol Prep Pads recall?
Call Cardinal Health at 1-800-292-9332 or email GMB-FieldCorrectiveAction@cardinalhealth.com. FDA’s Division of Industry and Consumer Education (DICE) can also help at 1-800-638-2041.
How do I report an infection potentially linked to Webcol™ Large Alcohol Prep Pads?
Submit a report through MedWatch or call 1-800-332-1088 to request a form.