FDA Recall: Revitaderm® & Tridergel™ Wound Care Gels Pulled for Bacterial Contamination
Blaine Labs, Inc. has announced a voluntary nationwide recall of select lots of Revitaderm® and Tridergel™ Wound Care Antiseptic Gel after routine quality testing detected the environmental bacterium Lysinibacillus fusiformis. Consumers, medical offices, and distributors should immediately stop using the affected products, verify lot numbers, and coordinate returns for a refund or replacement.
Reason for the Revitaderm® and Tridergel™ Recall
Laboratory tests revealed Lysinibacillus fusiformis contamination in three production lots of Blaine Labs’ 1-oz and 3-oz wound care gels. Although the organism is typically low in pathogenicity, it forms hardy spores that tolerate harsh conditions and may cause infections, delayed healing, or other complications—especially in patients with open wounds, diabetes, or compromised immune systems.
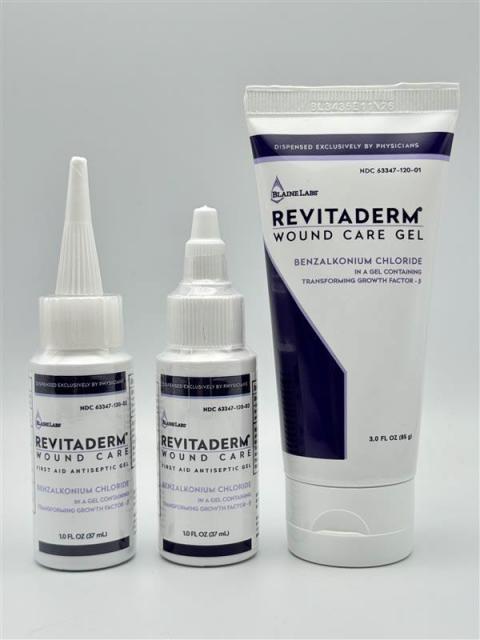
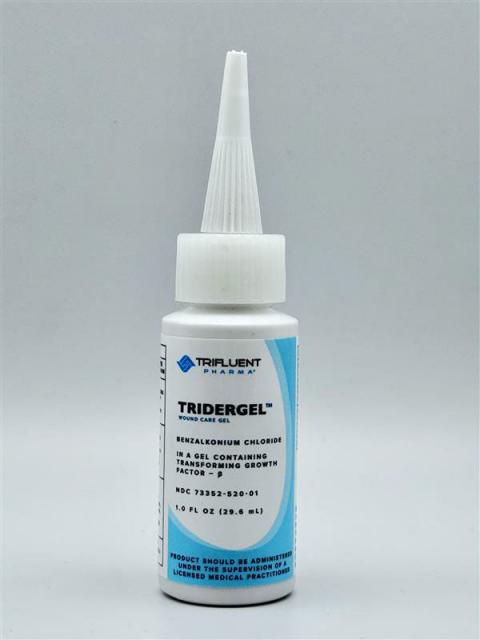
Recalled Product Details & Lot Numbers
- Revitaderm® Wound Care Antiseptic Gel – 1-oz bottle
- Lot BL3608 – Exp. 07/01/2028 (yellow ink, short twist cap)
- Lot BL3435 – Exp. 11/06/2026 (black ink, long pointy cap)
- Lot BL3525 – Exp. 08/07/2027 (black ink, long pointy cap)
- Revitaderm® Wound Care Antiseptic Gel – 3-oz tube
- Lot BL3435 – Exp. 11/06/2026 (imprinted on crimp)
- Lot BL3525 – Exp. 08/07/2027 (imprinted on crimp)
- Tridergel™ Wound Care Gel – 1-oz bottle
- Lot BL3435 – Exp. 11/06/2026 (black ink, long pointy cap)
Distribution Scope
The recalled gels were shipped nationwide within the United States directly to healthcare providers and physician offices. They were not sold through pharmacies, retail stores, or online marketplaces.
What Consumers & Clinics Should Do Now
- Locate any Revitaderm® or Tridergel™ Wound Care Gel in your facility or first-aid supplies.
- Verify the lot number on the bottle bottom (1-oz) or tube crimp (3-oz).
- Immediately discontinue use and quarantine any recalled lots.
- Contact Blaine Labs to arrange product return and reimbursement.
- Report adverse reactions or quality problems to the FDA MedWatch program.
Blaine Labs Recall Contact
Blaine Labs, Inc.
11037 Lockport Pl, Santa Fe Springs, CA 90670
Phone: (800) 307-8818
Email: Contact@blainelabs.com
Hours: Monday–Friday, 7:30 AM – 4:00 PM PST
Reporting to FDA MedWatch
Healthcare professionals and consumers can notify the FDA of any adverse events or quality concerns through MedWatch:
- Online: www.fda.gov/medwatch/report.htm
- Form Request: 1-800-332-1088
- Fax: 1-800-FDA-0178
Sources
Official Recall Images
Frequently Asked Questions
1. What should I do if I already applied the recalled Revitaderm® or Tridergel™ gel?
If your wound is healing normally and you are otherwise healthy, the infection risk is low. Monitor for redness, swelling, fever, or increased pain. Seek medical advice if any symptoms appear.
2. Are Revitaderm® or Tridergel™ products with different lot numbers safe to use?
Yes. Only the lot numbers listed in this recall are affected. Other lots have passed sterility testing and remain on the market.
3. How can I obtain a refund or replacement for recalled Revitaderm®/Tridergel™ gel?
Contact Blaine Labs at (800) 307-8818 or email Contact@blainelabs.com. The company will provide shipping instructions and issue reimbursement or replacement product.
4. Why weren’t Revitaderm® and Tridergel™ wound gels sold in pharmacies or online?
Blaine Labs distributes these professional-grade products exclusively to medical practices to ensure clinical oversight and appropriate wound-care use.
5. What infections can Lysinibacillus fusiformis from contaminated Revitaderm® or Tridergel™ cause?
The bacterium can lead to delayed wound healing, localized skin infections, or, in rare cases, bloodstream infections in immunocompromised individuals.
6. Do I need to notify patients treated with the recalled Revitaderm® or Tridergel™ lot?
Clinicians should review patient records, inform any affected individuals, and document follow-up care per standard infection-control protocols.
This independent report summarizes FDA recall information released on April 8, 2026. Always review the official FDA notice for full details. This site is not affiliated with Blaine Labs or the FDA.