FDA Recall: Amneal Magnesium Sulfate IV Bags Recalled for Potential Product Mix-Up
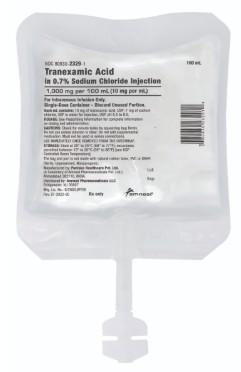
March 24 2026 — Bridgewater, NJ: Amneal Pharmaceuticals has issued a nationwide recall of one lot of its Magnesium Sulfate in Water for Injection (4 g/100 mL) IV bags after a hospital discovered a Tranexamic Acid bag sealed inside the outer pouch. Hospitals must immediately quarantine lot AH250162 and return the affected inventory — do not administer to patients.
Why the Amneal Magnesium Sulfate IV Recall Matters
- Critical-therapy delay: Pregnant patients with pre-eclampsia/eclampsia who do not receive magnesium in time are at heightened risk of seizures and neurological injury.
- Wrong-drug hazards: Inadvertent IV administration of Tranexamic Acid can cause thrombosis, seizures, hypersensitivity reactions and visual disturbances.
- No injuries reported (yet): Amneal has received no adverse-event reports so far, but the potential for patient harm is significant.
How to Identify the Recalled Amneal Magnesium Sulfate Lot
- Product: Magnesium Sulfate in Water for Injection, USP 4 g/100 mL IV bag
- National Drug Code (NDC): 70121-1720-3
- Affected lot: AH250162
- Packaging: 12 × 100 mL pouches per carton
- Distribution dates: 12/22/2025 – 02/27/2026 (U.S. nationwide)
Required Hospital Actions
- Immediately stop using and isolate any inventory from lot AH250162.
- Arrange a return to:
Amneal Pharmaceuticals – Magnesium Sulfate Recall Coordinator
21 Colonial Drive, Piscataway, NJ 08854 - Alert pharmacy and clinical staff; verify contents of every Magnesium Sulfate pouch in stock before dispensing.
- Report adverse events to FDA MedWatch:
- Online reporting form
- Fax: 1-800-FDA-0178 | Mail: form available on FDA website
- Questions? Contact Amneal:
- Phone: 833-582-0812 (Mon–Fri, 8 am–5 pm EST)
- Email: MagnesiumSulfateRecall@amneal.com
Primary Sources
Official Recall Images


Frequently Asked Questions
1. Why is Amneal recalling its Magnesium Sulfate IV bags?
A pouch labeled for Magnesium Sulfate was found to contain Tranexamic Acid, creating a life-threatening product mix-up risk.
2. Which lot numbers of Amneal Magnesium Sulfate IV bags are affected?
Only Lot AH250162 of Magnesium Sulfate in Water for Injection 4 g/100 mL is under recall.
3. What are the dangers if Tranexamic Acid is infused instead of Amneal Magnesium Sulfate?
Potential adverse events include thrombosis, seizures, hypersensitivity reactions, visual disturbances and dizziness.
4. How should hospitals return the recalled Amneal Magnesium Sulfate IV bags?
Ship the isolated product to Amneal Pharmaceuticals, 21 Colonial Drive, Piscataway, NJ 08854, Attn: Magnesium Sulfate Recall Coordinator, using standard return procedures.
5. Has the FDA classified the Amneal Magnesium Sulfate recall yet?
The recall is being conducted with FDA oversight, but the agency has not posted its official classification (Class I, II or III) as of this update.
6. Are home-use patients impacted by the Amneal Magnesium Sulfate IV bag recall?
No. The product is intended for hospital use only; retail consumers are not affected.
7. What should clinicians verify before administering an Amneal Magnesium Sulfate IV bag?
Confirm the pouch label, cross-check the inner bag’s printed drug name and inspect the solution for clarity before infusion.
8. How can clinicians or consumers contact Amneal about the Magnesium Sulfate IV recall?
Call 833-582-0812 (8 am–5 pm EST) or email MagnesiumSulfateRecall@amneal.com for recall logistics and medical information.